-Positive Interim Clinical Trial Data for the First Oculopharyngeal Muscular Dystrophy (OPMD) Subject Dosed with BB-301 in the Phase 1b/2a Clinical Treatment Study Reported in April-
-The Second OPMD Subject was Safely Dosed with BB-301 in February-
-Additional Interim Clinical Safety Data and Clinical Efficacy Data are Expected for Several Study Subjects in the Second Half of 2024-
-23 OPMD Subjects have Enrolled into the OPMD Natural History Study with Multiple Subjects Eligible for Entry into the BB-301 Phase 1b/2a Clinical Treatment Study-
-Closed an Oversubscribed Private Placement Financing of
“The interim clinical trial results for the first subject enrolled onto the BB-301 Phase 1b/2a Clinical Treatment Study demonstrated consistent, clinically meaningful improvements in each of the central study endpoints, with significant improvements noted across the radiographic assessments of swallowing and corresponding improvements observed for the key subject-reported outcome measure,” said
Operational Updates
On
The financing was led by
In connection with the PIPE financing, the Company has agreed with long-term investor
The key milestones related to the development of BB-301 for the treatment of OPMD-related Dysphagia, along with other corporate updates, are outlined below:
Interim BB-301 Phase 1b/2a Clinical Treatment Study Results for Subject 1:
- During the OPMD Natural History (NH) Study, which represents the pre-dose observational period for each subject, Subject 1 experienced progressive worsening of dysphagia as demonstrated by the results of the videofluoroscopic swallowing studies (VFSS), the cold water timed drinking test, and the key subject-reported outcome measure (the Sydney Swallow Questionnaire). VFSS represents the gold standard analytical method for the quantitative assessment of dysphagia (swallowing difficulty) in the clinical setting.
- At the 90-day post-dose assessment following the administration of BB-301, Subject 1 demonstrated improvements in key videofluoroscopic assessments which correlated with the observation of similar improvement in the key subject-reported outcome measure as compared to the average values for the respective assessments completed during the pre-dose observational period (as summarized in Figure 1). Notably, the results of many assessments completed at the 90-day timepoint demonstrated improvements over the initial measurements assessed at the subject’s first visit for the natural history observational study which occurred more than 12 months prior to the 90-day post-dose assessment.
- The most significant VFSS improvements at Day 90 were observed for swallowing tasks centered on the evaluation of pharyngeal constrictor muscle function and swallowing efficiency in the context of the consumption of thin liquids (e.g., water), solid foods, and thick non-solid foods (e.g., yogurt or pudding) (Figure 1). The VFSS improvements correlated with an improvement in the key subject-reported outcome measure (the Sydney Swallow Questionnaire), indicating an improvement in swallowing function as reported by Subject 1 (Figure 1).
Figure 1: Improvement in All Outcomes at 90-Days Post-BB-301 Injection*
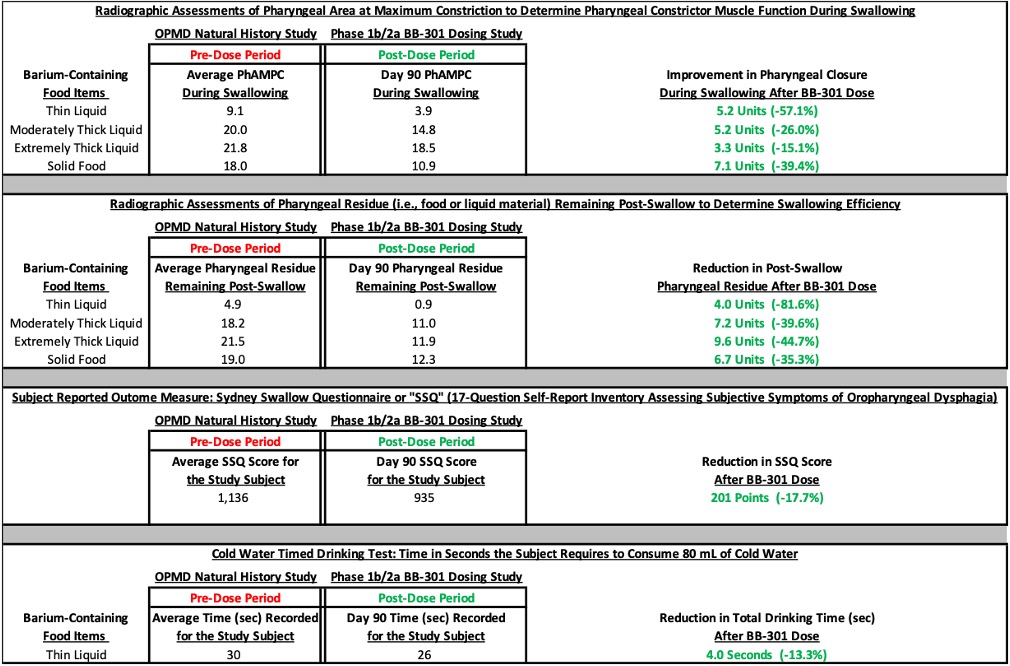
*Company data on file
- Regarding the BB-301 safety profile observed to date, no Serious Adverse Events (SAEs) have been observed for the two subjects that have received BB-301.
Transient Grade 2 Gastroesophageal Reflux Disease or “GERD” (i.e., “acid reflux” or “heartburn”) was observed for the two subjects that received BB-301. For both study subjects, the GERD resolved following the use of common prescription medications approved for the treatment of GERD.
Corporate Updates:
- Twenty-three (23) subjects have been enrolled into the OPMD NH Study, and the BB-301 Phase 1b/2a Clinical Treatment Study is designed to treat 21-to-30 subjects. Two (2) subjects have rolled over from the OPMD NH Study into the BB-301 Phase 1b/2a Clinical Treatment Study, and a third OPMD subject is expected to enter the BB-301 Phase 1b/2a Clinical Treatment Study in
June 2024 .
- Additional interim safety data and efficacy data are expected to be reported from the BB-301 Phase 1b/2a Clinical Treatment Study in 2H 2024.
Financial Highlights
Third Fiscal Quarter 2024 Financial Results
Total Revenues for the quarter ended
Total Expenses for the quarter ended
The loss from operations for the quarter ended
Consolidated Balance Sheets (in thousands, except par value and share amounts) | |||||||
| 2024 | 2023 | ||||||
| (Unaudited) | |||||||
| Assets | |||||||
| Current assets: | |||||||
| Cash at bank | $ | 14,143 | $ | 2,477 | |||
| Restricted cash | 13 | 13 | |||||
| Trade and other receivables | 53 | 55 | |||||
| Prepaid and other assets | 157 | 1,184 | |||||
| Total current assets | 14,366 | 3,729 | |||||
| Property and equipment, net | 204 | 87 | |||||
| Deposits | 25 | 25 | |||||
| Prepaid and other assets | 69 | 97 | |||||
| Right-of-use assets | 335 | 526 | |||||
| Total assets | $ | 14,999 | $ | 4,464 | |||
| Liabilities and stockholders’ equity | |||||||
| Current liabilities: | |||||||
| Trade and other payables | $ | 2,628 | $ | 3,231 | |||
| Accrued employee benefits | 517 | 472 | |||||
| Lease liabilities, current portion | 292 | 275 | |||||
| Total current liabilities | 3,437 | 3,978 | |||||
| Lease liabilities, less current portion | 62 | 284 | |||||
| Total liabilities | 3,499 | 4,262 | |||||
| Commitments and contingencies (Note 11) | |||||||
| Stockholders’ equity: | |||||||
| Common stock, | — | — | |||||
| Additional paid-in capital | 197,255 | 168,921 | |||||
| Accumulated deficit | (184,920 | ) | (167,889 | ) | |||
| Accumulated other comprehensive loss | (835 | ) | (830 | ) | |||
| Total stockholders’ equity | 11,500 | 202 | |||||
| Total liabilities and stockholders’ equity | $ | 14,999 | $ | 4,464 | |||
The accompanying notes are an integral part of these consolidated financial statements.
Consolidated Statements of Operations and Comprehensive Loss (Unaudited) (in thousands, except share and per share amounts) | |||||||||||||||
| Three Months Ended | Nine Months Ended | ||||||||||||||
| 2024 | 2023 | 2024 | 2023 | ||||||||||||
| Revenue: | |||||||||||||||
| Licensing revenues from customers | $ | — | $ | 54 | $ | — | $ | 68 | |||||||
| Total revenues | — | 54 | — | 68 | |||||||||||
| Operating expenses: | |||||||||||||||
| Royalties and license fees | (3 | ) | — | (108 | ) | — | |||||||||
| Research and development | 2,566 | 3,167 | 12,097 | 9,588 | |||||||||||
| General and administrative | 1,578 | 1,228 | 4,953 | 5,011 | |||||||||||
| Total operating expenses | 4,141 | 4,395 | 16,942 | 14,599 | |||||||||||
| Loss from operations | (4,141 | ) | (4,341 | ) | (16,942 | ) | (14,531 | ) | |||||||
| Other income (loss): | |||||||||||||||
| Foreign currency transaction loss | (118 | ) | (45 | ) | (22 | ) | (391 | ) | |||||||
| Interest expense, net | (4 | ) | (7 | ) | (16 | ) | (25 | ) | |||||||
| Other income (expense), net | (16 | ) | — | (50 | ) | 50 | |||||||||
| Unrealized loss on investment | — | (4 | ) | (1 | ) | (4 | ) | ||||||||
| Total other income (loss), net | (138 | ) | (56 | ) | (89 | ) | (370 | ) | |||||||
| Net loss | $ | (4,279 | ) | $ | (4,397 | ) | $ | (17,031 | ) | $ | (14,901 | ) | |||
| Other comprehensive income (loss): | |||||||||||||||
| Unrealized foreign currency translation gain (loss) | 117 | 45 | (5 | ) | 392 | ||||||||||
| Total other comprehensive income (loss) | 117 | 45 | (5 | ) | 392 | ||||||||||
| Total comprehensive loss | $ | (4,162 | ) | $ | (4,352 | ) | $ | (17,036 | ) | $ | (14,509 | ) | |||
| Net loss | $ | (4,279 | ) | $ | (4,397 | ) | $ | (17,031 | ) | $ | (14,901 | ) | |||
| Net loss per share: | |||||||||||||||
| Basic and diluted | $ | (1.64 | ) | $ | (2.67 | ) | $ | (6.95 | ) | $ | (11.47 | ) | |||
| Weighted average number of shares outstanding: basic and diluted | 2,616,288 | 1,645,951 | 2,449,295 | 1,299,423 | |||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
About BB-301
BB-301 is a novel, modified AAV9 capsid expressing a unique, single bifunctional construct promoting co-expression of both codon-optimized Poly-A Binding Protein Nuclear-1 (PABPN1) and two small inhibitory RNAs (siRNAs) against mutant PABPN1. The two siRNAs are modeled into microRNA backbones to silence expression of faulty mutant PABPN1, while allowing expression of the codon-optimized PABPN1 to replace the mutant with a functional version of the protein. We believe the silence and replace mechanism of BB-301 is uniquely positioned for the treatment of OPMD by halting mutant expression while providing a functional replacement protein.
About
Forward Looking Statements
Except for the historical information set forth herein, the matters set forth in this press release include forward-looking statements, including statements regarding Benitec’s plans to develop and commercialize its product candidates, the timing of the completion of pre-clinical and clinical trials, the timing of the availability of data from our clinical trials, the timing and sufficiency of patient enrollment and dosing in clinical trials, the timing of expected regulatory filings, the clinical utility and potential attributes and benefits of ddRNAi and Benitec’s product candidates, the intellectual property position, the length of time over which the Company expects its cash and cash equivalents to be sufficient to execute on its business plan, and other forward-looking statements.
These forward-looking statements are based on the Company’s current expectations and subject to risks and uncertainties that may cause actual results to differ materially, including unanticipated developments in and risks related to: unanticipated delays; further research and development and the results of clinical trials possibly being unsuccessful or insufficient to meet applicable regulatory standards or warrant continued development; the ability to enroll sufficient numbers of subjects in clinical trials; determinations made by the FDA and other governmental authorities and other regulatory developments; the Company’s ability to protect and enforce its patents and other intellectual property rights; the Company’s dependence on its relationships with its collaboration partners and other third parties; the efficacy or safety of the Company’s products and the products of the Company’s collaboration partners; the acceptance of the Company’s products and the products of the Company’s collaboration partners in the marketplace; market competition; sales, marketing, manufacturing and distribution requirements; greater than expected expenses; expenses relating to litigation or strategic activities; the Company’s ability to satisfy its capital needs through increasing its revenue and obtaining additional financing; the impact of the COVID-19 pandemic, the disease caused by the SARS-CoV-2 virus or any similar events, which may adversely impact the Company’s business and pre-clinical and clinical trials; the impact of local, regional, and national and international economic conditions and events; and other risks detailed from time to time in the Company’s reports filed with the
Investor Relations Contact:
(917) 734-7387
ikoffler@lifesciadvisors.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/8bb63807-b739-42f2-a124-8caa83944152

![]()
Figure One

Improvement in All Outcomes at 90-Days Post-BB-301 Injection*
2024 GlobeNewswire, Inc., source







