IRVINE, CA, Feb. 25, 2019 (GLOBE NEWSWIRE) --
| |||||
Pre-Clinical Work and Animal Studies Underway, and Protocols for Clinical Studies Under Development
via NEWMEDIAWIRE -- Cannabis Science, Inc. (OTC: CBIS), a U.S. company specializing in the development of cannabinoid-based medicines, announces that it intends to expand the scope of its sponsored research in Nigeria to include neurological conditions, including Post-Traumatic Stress Disorder (PTSD). CBIS and its research collaborator are currently investigating the potential beneficial effects of cannabidiol on oro-bucco-lingual dyskinesias, oxidative stress, and psychosis using animal studies and a clinical trial.
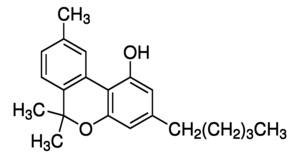
CBIS recently announced receipt of U.S. Patent Number 9,763,991 for Compositions of Cannabinol (CBN) for Treatment of Various Neurobehavioral Disorders, Sleep Deprivation (Insomnia), Anxiety Disorders, and PTSD, among other target indications.
The invention relates to a composition for use in the treatment of neurobehavioral disorders utilizing various Cannabis plant extracts comprising CBN preferably with other constituents of this plant for such use and a method for the extraction of plants. The plants or plant parts may for instance be derived from Cannabis Sativa and/or Cannabis Indica and/or Cannabis ruderalis and/or other elements, and mixtures thereof. The plant extracts derived showed particularly beneficial effects against Sleeping Disorders, in particular Insomnia, PTSD, and anxiety disorders including Attention Deficit Hyperactivity Disorder (ADHD).
The animal studies are well underway and should be completed in the coming months. This study is important in a country like Nigeria where movement disorders are still common because the relatively inexpensive conventional antipsychotics are still widely in use. Movement disorders are known to be more common in patients on the conventional antipsychotics, though they have also been reported in the atypical antipsychotics. It is therefore pertinent to continue to search for new and better forms of management of this difficult to manage movement disorder in psychotic patients.
CBIS has entered into an initial, one-year Research Collaboration Agreement with Stellenbosch University. CBIS and Stellenbosch University intend to jointly develop and investigate the use of Cannabinoids to treat chronic pelvic pain disorder/chronic prostatitis, and potentially other indications. Stellenbosch University (SU) is one of the oldest Universities in South Africa. The research is being conducted in South Africa and Nigeria.
Cannabis Science is aggressively expanding the Company’s network of research centers. Negotiations are ongoing with several academic institutions in the United States and internationally to establish new, collaborative research agreements. These contracts will focus on several indications, and are expected to expand the research funded, and directed by the Company. In addition, Cannabis Science will develop multi-center, clinical trial networks with cannabinoid drug development.
“We are encouraged with the preliminary results from our pre-clinical work and animal studies in Nigeria,” stated Mr. Raymond C. Dabney, CBIS’ President, CEO, and Co-founder. “The research may be of use in other movement disorders, such as Huntington’s Disease, Tourette’s Syndrome, Cerebral Palsy, and Dystonia. These could be related through the neurotransmitter, dopamine, modulated by the CB1 receptors. While we will continue our research in regard to oro-bucco-lingual dyskinesias, oxidative stress, and psychosis, we believe there is an opportunity to expand our research in Nigeria to include PTSD. Specifically, we will develop a protocol for a PTSD clinical study immediately and begin this study as soon as possible. This will allow the Company to leverage our patent for compositions of cannabinol (CBN), as well as our expertise with other cannabinoids.”
“Our work in Nigeria, while independent of our research collaborations in the U.S., contributes significantly to the Global Consortium we launched last year,” added Mr. Dabney. The Cannabis Science Global Consortium provides the Company with a framework to cooperate and collaborate with stakeholders worldwide. The objective of the consortium is to harness the collective expertise of CBIS’ global partners to investigate the use of Cannabinoids for the treatment of a variety of indications. The consortium links universities, foundations, corporations, and individuals to share research, ideas, and other relevant information, as well as to implement a cutting-edge research program to develop medicines and delivery mechanisms from bench-to-bedside.
There are three primary elements in Cannabis Science’s drug development program: (1) Defining the effectiveness and specificity of cannabinoids being tested; (2) Identifying and testing drug delivery mechanisms; and (3) Testing and evaluating the impact of co-interventions, and the application of additional diagnostic and/or therapeutic procedures to participants in our randomized controlled trials. The Company’s initial results point to the importance of drug delivery systems and the impact of co-interventions.
About Stellenbosch University
Stellenbosch University (SU) is one of the oldest Universities in South Africa. SU is home to an academic community of 29,000 students (including 4,000 foreign students from 100 countries) as well as 3,000 permanent staff members (including 1,000 academics) on five campuses. SU’s Department of Psychiatry is committed, through research, to better the understanding of psychiatric disorders in South Africa and beyond. SU seeks out research partnerships within the university and with other local and international institutions to harness available expertise and enhance the dissemination of SU’s findings as reflected on SU’s publications over the last several years. SU runs a thriving research program involving both basic and clinical studies focusing on schizophrenia, anxiety disorders, posttraumatic stress disorder, fetal alcohol syndrome, neurodegenerative disorders, neuroAIDS, substance abuse, eating disorders and the intersection of psychiatric and other non-communicable disorders. SU’s research is directly geared at addressing the disease burden in South Africa.
About Cannabis Science, Inc.
Cannabis Science’s primary objective is to research and develop U.S. Food and Drug Administration (FDA)-approved, cannabinoid-based medicine to fight a number of targeted critical ailments, including various Cancers, Neurological Conditions, PTSD, Sleep Deprivation, Chronic Pain, HIV/AIDS, Autism, Parkinson’s Disease, Epilepsy, Chronic Obstructive Pulmonary Disease (COPD), and others. CBIS and its collaborators are well underway on a number of research projects targeting Lung Cancer, Pancreatic Cancer, Chronic Pelvic Pain, and Oro-bucco-lingual Dyskinesias.
Cannabis Science, Inc. takes advantage of its unique understanding of metabolic processes to provide novel treatment approaches to a number of illnesses for which current treatments and understanding remain unsatisfactory. Cannabinoids have an extensive history dating back thousands of years, and currently, there are a growing number of peer-reviewed scientific publications that document the underlying biochemical pathways that cannabinoids modulate. The Company works with leading experts in drug development, medicinal characterization, and clinical research to develop, produce, and commercialize novel therapeutic approaches for the treatment for illnesses caused by infections as well as for age-related illness. Our initial focus is on cancers, HIV/AIDS, and neurological conditions. The Company is proceeding with the research and development of its proprietary drugs as a part of this initial focus: CS-S/BCC-1, CS-TATI-1, and CS-NEURO-1, respectively.
Forward-Looking Statements
This Press Release includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Act of 1934. A statement containing words such as "anticipate," "seek," intend," "believe," "estimate," "expect," "project," "plan," or similar phrases may be deemed "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Some or all of the events or results anticipated by these forward-looking statements may not occur. Factors that could cause or contribute to such differences include the future U.S. and global economies, the impact of competition, and the Company's reliance on existing regulations regarding the use and development of cannabis-based drugs. Cannabis Science, Inc., does not undertake any duty nor does it intend to update the results of these forward-looking statements. Safe Harbor Statement. The Private Securities Litigation Reform Act of 1995 provides a 'safe harbor' for forward looking statements. Certain of the statements contained herein, which are not historical facts are forward looking statements with respect to events, the occurrence of which involved risks and uncertainties. These forward-looking statements may be impacted, either positively or negatively, by various factors. Information concerning potential factors that could affect the company is detailed from time to time in the company's reports filed with the Securities and Exchange Commission.
Attachment
- CBN
Contact Information Cannabis Science, Inc. Mr. Raymond C. Dabney President & CEO, Co-Founder info@cannabisscience.com Tel: 1-888-263-0832
![]()