Vitiligo causes the progressive loss of skin pigment in lesions (patches) across the body and affects an estimated 45 million individuals worldwide. Pigment loss caused by vitiligo is most pronounced in patients with darker skin types who report the greatest impact of the disease on their quality of life. Treatment options remain limited, with no treatments for repigmentation of vitiligo currently approved by the
“Many vitiligo patients must grapple with a sense of lost identity as their skin visibly and inexplicably changes, forcing them to withdraw from society and having a severe impact on their mental health and quality of life,” CLINUVEL’s Director of North American Operations, Dr
“Clinical trials of afamelanotide to date have shown the drug can assist in repigmenting vitiligo patients’ skin, with the most promising results in darker skinned patients. Therefore we have chosen to focus on this patient group, with the highest unmet need, as our clinical program progresses,”
Afamelanotide as repigmentation therapy
One of a new class of drugs known as melanocortins, afamelanotide activates melanin, the pigment in skin. The drug is also understood to play a role in maintaining the overall health of skin including melanocytes, the skin cells responsible for melanin production.
“Many experimental vitiligo treatment approaches to date have been ineffective, inconvenient, or cause unpleasant side effects, often seeking to bluntly suppress the immune system to allow for repigmentation,”
CLINUVEL’s initial studies in vitiligo patients showed that afamelanotide could repigment skin in combination with a light-based therapy, with nearly 100 patients receiving treatment to date and the safety profile of the drug maintained.1,2 The program will now focus on afamelanotide as a monotherapy after approval of the CUV104 study was received from the
CUV104 will commence in
Up to six patients will be enrolled in CUV104, sufficient to meet the statistical needs of a pilot study, provide feedback on protocol designs for larger studies of afamelanotide as a monotherapy, and complete quickly in a clinical environment where COVID-19 restrictions have impacted study enrolment rates.
In total, over 11,000 doses of afamelanotide have been administered to over 1,600 individuals worldwide across clinical, special access and post-authorisation programs.2
Media enquiries:
1 Results from these studies have been published in peer-reviewed journals:
- Lim, H. W. et al. (2015). Afamelanotide and Narrowband UV-B Phototherapy for the Treatment of Vitiligo: A Randomized Multicenter Trial. JAMA Dermatology, 151(1), 42.
- Toh, J. et al. (2020). Afamelanotide implants and narrow-band ultraviolet B phototherapy for the treatment of nonsegmental vitiligo in Asians.
Journal of the American Academy of Dermatology , 82(6), 1517–1519.
2 SCENESSE® (afamelanotide 16mg) is approved in the
About
CLINUVEL (ASX: CUV;
CLINUVEL’s lead therapy, SCENESSE® (afamelanotide 16mg), is approved for commercial distribution in
SCENESSE®, PRÉNUMBRA®, and NEURACTHEL® are registered trademarks of CLINUVEL.
Head of Investor Relations
Mr
Investor Enquiries
https://www.clinuvel.com/investors/contact-us
Forward-Looking Statements
This release contains forward-looking statements, which reflect the current beliefs and expectations of CLINUVEL’s management. A complete copy of our forward-looking statements advisory is available on www.clinuvel.com.
www.clinuvel.com Level 11,
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/3966307a-d28b-479f-a7cd-51ba609a84b8


Vitiligo repigmentation in CLINUVEL's CUV102 study
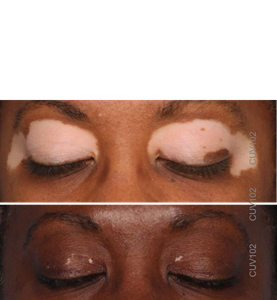
Initial clinical studies of afamelanotide showed the drug could repigment skin. This patient received four afamelanotide implants and NB-UVB light therapy over six months, largely repigmenting lesions on their eyelids. Images taken at days 0 and 171 of CUV102 study, courtesy of the study investigator.
2022 GlobeNewswire, Inc., source


