| ||||||||||
Note to editors: CLINUVEL is releasing in-depth infographics and a video explaining the mechanisms of action in XP and the overall DNA repair process. For updates, follow:
Twitter | Facebook | Instagram | LinkedIn
In the search for a preventative treatment of skin cancers, including melanoma, it is imperative to understand and treat DNA damage caused by ultraviolet (UV) radiation. Following the treatment of a patient suffering from xeroderma pigmentosum (XP), a disease characterised by an inborn insufficiency to repair DNA damaged by sun exposure, Australian based
CLINUVEL’s drug SCENESSE® (afamelanotide 16mg) belongs to a group of hormones which have been shown to reduce UV-induced damage to DNA (photoproducts) and assist in DNA regeneration.1 The Company is running a staged clinical program to confirm the ability of SCENESSE® to repair the DNA in skin cells, focusing initially on XP.
SCENESSE® IN XERODERMA PIGMENTOSUM
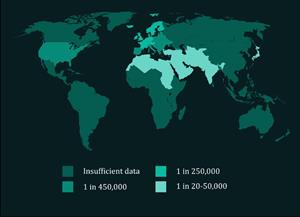
XP is a genetic disease which has served as a human model for studying the insufficiency of human DNA repair. Patients develop frequent skin cancers from an early age – most experience their first malignancy before adolescence – and must avoid all forms of UV exposure. The disease has a high mortality rate, with a median life expectancy of thirty years. XP treatment is limited to management of symptoms, in particular regular surgery to remove cancerous lesions. An estimated 1 in 450,000 individuals in
Today CLINUVEL announced that SCENESSE® has been administered for the first time to a patient diagnosed with XP under a Special Access Program, whereby the patient’s safety will be evaluated over six weeks of treatment. Following confirmation of safety of the drug product in this patient, CLINUVEL will conduct two further studies as part of the DNA Repair Program. Both studies – an open-label Phase II study involving six XP-C patients (CUV150) and a control study enrolling 10 healthy volunteers (CUV151) – will evaluate the impact of treatment with SCENESSE® on DNA damage and restoration.
Commentary
“We seek to provide meaningful benefit to XP patients, and these results will serve a wider population of fair-skinned individuals at risk of developing skin cancers,” CLINUVEL’s Clinical Operations Manager, Dr
CLINUVEL is developing a range of products based on its very targeted research with SCENESSE®, including topical formulations of its proprietary drugs and over-the-counter products, and has recently opened a new
1 SCENESSE® (afamelanotide 16mg) is approved in the
Authorised for ASX release by the Board of Directors of
About
SCENESSE® and PRÉNUMBRA® are registered trademarks of
Media enquiries
Mr
Level 39,
Head of Investor Relations
Mr
Investor enquiries
https://www.clinuvel.com/investors/contact-us
Forward-Looking Statements
This release contains forward-looking statements, which reflect the current beliefs and expectations of CLINUVEL’s management. Statements may involve a number of known and unknown risks that could cause our future results, performance or achievements to differ significantly from those expressed or implied by such forward-looking statements. Important factors that could cause or contribute to such differences include risks relating to: our ability to develop and commercialise pharmaceutical products, the COVID-19 pandemic affecting the supply chain for a protracted period of time, including our ability to develop, manufacture, market and sell biopharmaceutical products; competition for our products, especially SCENESSE® (afamelanotide 16mg); our ability to achieve expected safety and efficacy results through our innovative R&D efforts; the effectiveness of our patents and other protections for innovative products, particularly in view of national and regional variations in patent laws; our potential exposure to product liability claims to the extent not covered by insurance; increased government scrutiny in either
| www.clinuvel.com | |
| Level 11 | T +61 3 9660 4900 |
| F +61 3 9660 4999 | |
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/e162ca4b-27bb-4716-97e5-49a603bbebda
https://www.globenewswire.com/NewsRoom/AttachmentNg/0d2e4b7a-f3b5-4efc-9d67-58ae0b7d3810

2020 GlobeNewswire, Inc., source