Regulatory News:
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20200721005606/en/
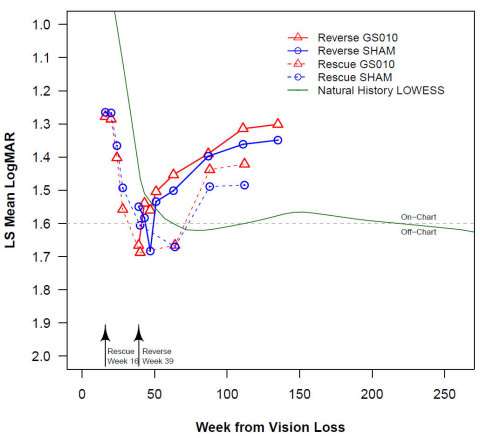
Figure 1. Evolution of Best-Corrected Visual Acuity (BCVA): REALITY Subjects (≥15 Years Old) with Mutated ND4 Gene vs. RESCUE and REVERSE Subjects. Note: BCVA = best-corrected visual acuity. The LOWESS line for REALITY (n=23 subjects aged 15 or older at time of vision loss) is based on a series of polynomial regressions around each data point. The regressions use a limited look back and look forward and give distant points less weight. The time course of BCVA for REVERSE and RESCUE uses the least-squares mean based on a mixed model ANCOVA analysis. The starting points of the curves are set to the average time from onset to time of treatment (16 weeks for RESCUE, 39 weeks for REVERSE).
GenSight Biologics (Paris:SIGHT) (Euronext: SIGHT, ISIN: FR0013183985, PEA-PME eligible), a biopharma company focused on discovering and developing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders, today reported results from the final analysis of the REALITY natural history study, which reaffirm the poor prognosis for the vast majority of Leber Hereditary Optic Neuropathy (LHON) patients with vision loss due to a mutated ND4 mitochondrial gene.
“The REALITY findings bolster the case that the improvements we saw in REVERSE, RESCUE and the first analysis of CLIN06 could not have arisen simply from spontaneous recovery among LHON patients,” commented Bernard Gilly, Co-founder and Chief Executive Officer of GenSight. “We intend to bring this and other scientific evidence to the authorities to make a robust and convincing argument about the outstanding therapeutic benefit delivered by LUMEVOQ®.”
LUMEVOQ® (GS010), the gene therapy developed by GenSight Biologics for the treatment of vision loss in patients with LHON caused by a mutated ND4 mitochondrial gene, is on track to be submitted for European marketing authorization in September 2020.
Statistical analysis of the visual acuity in 23 REALITY subjects aged 15 or older with a mutated ND4 gene shows that on average, vision failed to recover from an initial sudden decline, even several years after vision loss. The sharp deterioration followed by an extended period of low visual acuity stands in sharp contrast with the improvements observed in the LUMEVOQ® RESCUE and REVERSE trials.
“The REALITY results are entirely in keeping with a recent meta-analysis currently in press in the Journal of Neuro-Ophthalmology*, which showed that only about 11% of ND4 LHON patients have some degree of meaningful spontaneous improvement in vision,” noted Dr. Nancy Newman, LeoDelle Jolley Professor of Ophthalmology and Neurology at the Emory University School of Medicine in Atlanta, GA, USA, and one of the leading authorities on LHON.
REALITY was a retrospective and cross-sectional observational study of subjects with LHON, conducted in centers across Spain, Italy, France, United Kingdom, and the United States. The objective was to generate insights about the natural history of the disease based on an approach that would facilitate comparisons with REVERSE and RESCUE. The study aimed to enroll 50 subjects, but enrollment was curtailed due to measures taken to protect patients in the COVID-19 pandemic.
Of the 44 subjects who were enrolled before the COVID-19 measures took effect, 23 were 15 years old or older at the time of vision loss due to their mutated ND4 gene. Of these 23 ND4 subjects, 15 had been treated with idebenone, the majority within 12 months of their vision loss.
The RESCUE and REVERSE pivotal trials evaluated the efficacy and safety of a single intravitreal injection of LUMEVOQ® in LHON subjects who were at 0-6 months and 6-12 months, respectively, from onset of vision loss due to carrying a mutated ND4 mitochondrial gene. 61 of the RESCUE and REVERSE subjects accepted the invitation for long-term follow-up in the CLIN06 trial, which recently reported initial results.
*N.J. Newman, V. Carelli, M. Taiel, P. Yu-Wai-Man. Visual outcomes in Leber hereditary optic neuropathy patients with the m. 11778>A (MTND4) mitochondrial DNA mutation. J Neuroophthalmol. In press (2020)
About GenSight Biologics
GenSight Biologics S.A. is a clinical-stage biopharma company focused on discovering and developing innovative gene therapies for retinal neurodegenerative diseases and central nervous system disorders. GenSight Biologics’ pipeline leverages two core technology platforms, the Mitochondrial Targeting Sequence (MTS) and optogenetics, to help preserve or restore vision in patients suffering from blinding retinal diseases. GenSight Biologics’ lead product candidate, GS010, is in Phase III trials in Leber Hereditary Optic Neuropathy (LHON), a rare mitochondrial disease that leads to irreversible blindness in teens and young adults. Using its gene therapy-based approach, GenSight Biologics’ product candidates are designed to be administered in a single treatment to each eye by intravitreal injection to offer patients a sustainable functional visual recovery.
About LUMEVOQ® (GS010)
LUMEVOQ® (GS010) targets Leber Hereditary Optic Neuropathy (LHON) by leveraging a mitochondrial targeting sequence (MTS) proprietary technology platform, arising from research conducted at the Institut de la Vision in Paris, which, when associated with the gene of interest, allows the platform to specifically address defects inside the mitochondria using an AAV vector (Adeno-Associated Virus). The gene of interest is transferred into the cell to be expressed and produces the functional protein, which will then be shuttled to the mitochondria through specific nucleotidic sequences in order to restore the missing or deficient mitochondrial function. “LUMEVOQ” was accepted as the invented name for GS010 (lenadogene nolparvovec) by the European Medicines Agency (EMA) in October 2018.
About Leber Hereditary Optic Neuropathy (LHON)
Leber Hereditary Optic Neuropathy (LHON) is a rare maternally inherited mitochondrial genetic disease, characterized by the degeneration of retinal ganglion cells that results in brutal and irreversible vision loss that can lead to legal blindness, and mainly affects adolescents and young adults. LHON is associated with painless, sudden loss of central vision in the 1st eye, with the 2nd eye sequentially impaired. It is a symmetric disease with poor functional visual recovery. 97% of patients have bilateral involvement at less than one year of onset of vision loss, and in 25% of cases, vision loss occurs in both eyes simultaneously. The estimated incidence of LHON is approximately 1,400 to 1,500 new patients who lose their sight every year in the United States and Europe.
About RESCUE and REVERSE
RESCUE and REVERSE are two separate randomized, double-masked, sham-controlled Phase III trials designed to evaluate the efficacy of a single intravitreal injection of GS010 (rAAV2/2-ND4) in subjects affected by LHON due to the G11778A mutation in the mitochondrial ND4 gene.
The primary endpoint will measure the difference in efficacy of GS010 in treated eyes compared to sham-treated eyes based on Best‑Corrected Visual Acuity (BCVA), as measured with the ETDRS at 48 weeks post-injection. The patients’ LogMAR (Logarithm of the Minimal Angle of Resolution) scores, which are derived from the number of letters patients read on the ETDRS chart, will be used for statistical purposes. Both trials have been adequately powered to evaluate a clinically relevant difference of at least 15 ETDRS letters between treated and untreated eyes adjusted to baseline.
The secondary endpoints will involve the application of the primary analysis to best‑seeing eyes that received GS010 compared to those receiving sham, and to worse‑seeing eyes that received GS010 compared to those that received sham. Additionally, a categorical evaluation with a responder analysis will be evaluated, including the proportion of patients who maintain vision (< ETDRS 15L loss), the proportion of patients who gain 15 ETDRS letters from baseline and the proportion of patients with Snellen acuity of >20/200. Complementary vision metrics will include automated visual fields, optical coherence tomography, and color and contrast sensitivity, in addition to quality of life scales, bio‑dissemination and the time course of immune response. Readouts for these endpoints are at 48, 72 and 96 weeks after injection.
The trials are conducted in parallel, in 37 subjects for REVERSE and 39 subjects for RESCUE, in 7 centers across the United States, the UK, France, Germany and Italy. Week 96 results were reported in 2019 for both trials, after which patients were transferred to a long-term follow-up study that will last for three years.
ClinicalTrials.gov Identifiers:
REVERSE: NCT02652780
RESCUE: NCT02652767
About CLIN06 (RESCUE and REVERSE Long-term Follow-up)
CLIN06 is the long-term follow-up study of ND4 LHON subjects treated with LUMEVOQ® (GS010) gene therapy in the RESCUE or REVERSE Phase III Clinical Trials. The total study period for an individual subject is 3 years, i.e., 5 years post-gene therapy administration. No study treatment is administered during CLIN06.
The primary objective isto assess the long-term safety of intravitreal LUMEVOQ® administration up to 5 years post-treatment. The secondary objective isto assess the long-term treatment efficacy of the therapy and the quality of life (QoL) in subjects up to 5 years post-treatment. The first subject was enrolled on January 9, 2018. 61 subjects have enrolled.
ClinicalTrials.gov Identifiers:
CLIN06: NCT03406104
About REALITY
REALITY is a multi-country retrospective and cross-sectional observational study of affected LHON subjects, based on subjects’ medical charts and the administration of surveys on Health-Related Quality of Life (HRQoL) and direct and indirect costs associated with the disease.
The study will recruit at least 50 subjects (both adult and pediatric) chiefly in the following countries: Spain, Italy, France, United Kingdom, and the United States.
The primary objectives for the REALITY study are: to describe the evolution of visual functional and structural changes and other associated symptoms in patients with LHON; understand the impact of LHON-related vision loss on the HRQoL; and understand the economic burden for patients and their families arising from direct and indirect costs associated with the disease. The secondary objective is to describe the relationship between genetic, lifestyle and/or environmental factors and the expression of the LHON phenotype.
The first subject was enrolled on 3 January 2018, and enrollment was completed in early Q2 2020.
ClinicalTrials.gov Identifiers:
REALITY LHON Registry: NCT03295071
View source version on businesswire.com: https://www.businesswire.com/news/home/20200721005606/en/


