- First collaboration program, leveraging Cyrus’ de novo computational approach to create proprietary IL-2 protein agonist targeting autoimmune and other deleterious immune conditions-
-Preclinical data demonstrate synergistic activity of ImmTOR in combination with IL-2 mutein to increase the level and durability of Treg expansion-
- Unlocking a new generation of innovative biologic therapeutics with the potential to improve the lives of patients-
WATERTOWN, Mass. and SEATTLE, Sept. 08, 2021 (GLOBE NEWSWIRE) -- Selecta Biosciences, Inc. (NASDAQ: SELB), a biotechnology company leveraging its clinically validated ImmTOR™ platform to develop tolerogenic therapies that selectively mitigate unwanted immune responses, and Cyrus Biotechnology, Inc. a leading protein design company, today announced a protein engineering collaboration combining Selecta’s ImmTOR platform with Cyrus’ ability to radically redesign protein therapeutics. The lead program in the collaboration is a proprietary interleukin-2 (IL-2) protein agonist designed to selectively promote expansion of regulatory T cells (Treg) for the treatment of patients with autoimmune diseases and other deleterious immune conditions.
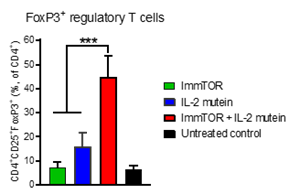
Novel IL-2 approaches and technologies are driving innovation in the therapeutic development space. The IL-2 pathway influences critical aspects of both immune stimulation and immune regulation, through the development and expansion of regulatory T cells (Treg). These Treg cells are a specialized subpopulation of T cells involved in suppressing certain immune responses and maintaining the body’s self-tolerance. Reductions in the number of Treg cells have been shown to drive a spectrum of autoimmune diseases and conversely, increasing Treg expansion may have clinical utility in reducing inflammation and improving disease outcomes. Early preclinical data investigating the effects of ImmTOR in combination with a Treg-selective IL-2 mutant protein (IL-2 “mutein”) demonstrate substantial synergistic activity in increasing the percentage and durability of Treg expansion in the spleen (Figure below). This supports the potential of ImmTOR in combination with IL-2 proteins to restore immunotolerance to autoantigens and forms the basis for this partnership. Although IL-2 has been an attractive target for autoimmune indications, overcoming its immunostimulatory activities, short half-life and anti-IL-2 antibody formation has been challenging. Building on recent advancements in the field, our strong preliminary data suggest that ImmTOR in combination with Cyrus’ novel IL-2 protein agonist has the potential to unlock the value of this target and drive the development of a next generation, best-in-class asset.
A figure accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/eb50b792-d824-4b4b-8e60-c2135fe26e2e
“We are excited to collaborate with Cyrus, and we see this strategic protein engineering partnership as an important step in advancing our ImmTOR platform for the treatment of autoimmune diseases,” said Carsten Brunn, Ph.D., President and Chief Executive Officer of Selecta. “We are encouraged by the preclinical data generated to date and the growing literature that supports the potential of IL-2 therapeutics in treating immunological diseases.”
Dr. Brunn added, “We look forward to leveraging Cyrus’ ability to both reengineer immune epitopes and rationally design novel proteins using non-traditional starting sequences, including non-human, non-natural and ancestral versions. For our first program, this combination will allow us to potentially mitigate unwanted immune responses by reducing the inherent immunogenicity of the protein while also promoting immune tolerance. We are fortunate to have this opportunity to optimize and advance our portfolio through the design and generation of innovative protein therapeutics. Beyond leading a paradigm shift in the way biologics are made, ultimately this collaboration has the potential to unlock new treatment options and improve the lives of patients who suffer from serious and debilitating diseases.”
Lucas Nivon, Ph.D., Chief Executive Officer of Cyrus, commented, “We welcome Selecta as a deeply experienced partner. This collaboration is in perfect alignment with our protein design expertise and represents an important endorsement of our platform, which has the potential to further enhance the potency of ImmTOR’s tolerizing power. With our current partners, we have demonstrated our ability to redesign existing protein biologics or build them from the ground-up -- expanding the potential for therapeutics and intellectual property. We look forward to executing on our shared vision.”
Under the terms of the collaboration, Selecta has provided an upfront payment, and Cyrus is eligible to earn discovery, development and sales based milestones. Novel engineered protein therapeutic candidates from the partnership will be used to expand Selecta’s proprietary pipeline and further bolster Selecta’s clinically validated ImmTOR platform.
About Selecta Biosciences, Inc.
Selecta Biosciences Inc. (NASDAQ: SELB) is a clinical stage biotechnology company leveraging its ImmTOR™ platform to develop tolerogenic therapies that selectively mitigate unwanted immune responses. With a proven ability to induce tolerance to highly immunogenic proteins, ImmTOR has the potential to amplify the efficacy of biologic therapies, including redosing of life-saving gene therapies, as well as restore the body’s natural self-tolerance in autoimmune diseases. Selecta has several proprietary and partnered programs in its pipeline focused on enzyme therapies, gene therapies, and autoimmune diseases. Selecta Biosciences is headquartered in the Greater Boston area. For more information, please visit www.selectabio.com.
About Cyrus Biotechnology
Cyrus Biotechnology is a pre-clinical-stage biotechnology company applying leading computational protein design capabilities coupled with massively parallel in vitro screening to engineer novel biologics drugs. Cyrus is advancing a novel pre-clinical infectious disease therapeutic and developing a pipeline of internal and partnered discovery programs over a range of indications, including next-generation CRISPR therapeutics in collaboration with the Broad Institute. Cyrus founders include Dr. David Baker of the University of Washington and the company’s platform is based on core software from the Baker lab. The company has worked with over 100 industry partners, including 13 of the top 20 global Pharma firms. Cyrus is based in Seattle, WA and financed by leading Biotech and Tech investors including Orbimed, Trinity, Springrock, WRF and Alexandria. For more information please visit cyrusbio.com.
Selecta Forward-Looking Statements
Any statements in this press release about the future expectations, plans and prospects of Selecta Biosciences, Inc. (“the Company”), including without limitation, statements regarding the unique proprietary technology platform of the Company, and the unique proprietary platform of its partners, the programs and disease indication targets anticipated under this collaboration, the potential of our partners to develop or redesign any protein therapeutics, the ability of any drug candidate developed under the collaboration to offer a therapeutic benefit, including the promotion of expansion of regulatory T cells for the treatment of autoimmune disease indications, the potential of ImmTOR to enable re-dosing of AAV gene therapy and to mitigate immunogenicity, the potential of ImmTOR and the Company’s product pipeline to treat chronic refractory gout, IgA nephropathy, PBC, MMA or OTC, the anticipated timing or the outcome of ongoing and planned clinical trials, studies and data readouts, the Company’s ability to conduct those clinical trials and studies, the timing or making of any regulatory filings, the potential treatment applications of product candidates utilizing the ImmTOR platform in areas such as gene therapy, gout and autoimmune disease, the ability of the Company and its partners where applicable to develop gene therapy products using ImmTOR, the novelty of treatment paradigms that the Company is able to develop, whether the observations made in non-human primate study subjects will translate to studies performed with human beings, the potential of any therapies developed by the Company to fulfill unmet medical needs, the Company’s plan to apply its ImmTOR technology platform to a range of biologics for rare and orphan genetic diseases, the potential of the Company’s intellectual property to enable repeat administration in gene therapy product candidates and products, the ability to re-dose patients and the potential of ImmTOR to allow for re-dosing, the potential to safely re-dose AAV, the ability to restore transgene expression, the potential of the ImmTOR technology platform generally and the Company’s ability to grow its strategic partnerships, and other statements containing the words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “hypothesize,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “would,” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including, but not limited to, the following: the uncertainties inherent in the initiation, completion and cost of clinical trials including proof of concept trials, including the uncertain outcomes, the availability and timing of data from ongoing and future clinical trials and the results of such trials, whether preliminary results from a particular clinical trial will be predictive of the final results of that trial or whether results of early clinical trials will be indicative of the results of later clinical trials, the ability to predict results of studies performed on human beings based on results of studies performed on non-human primates, the unproven approach of the Company’s ImmTOR technology, our partners’ ability to re-engineer or develop any protein therapeutics, potential delays in enrollment of patients, undesirable side effects of the Company’s product candidates, its reliance on third parties to manufacture its product candidates and to conduct its clinical trials, the Company’s inability to maintain its existing or future collaborations, licenses or contractual relationships, its inability to protect its proprietary technology and intellectual property, potential delays in regulatory approvals, the availability of funding sufficient for its foreseeable and unforeseeable operating expenses and capital expenditure requirements, the Company’s recurring losses from operations and negative cash flows from operations raise substantial doubt regarding its ability to continue as a going concern, substantial fluctuation in the price of its common stock, and other important factors discussed in the “Risk Factors” section of the Company’s most recent Quarterly Report on Form 10-Q to be filed after this release, and in other filings that the Company makes with the Securities and Exchange Commission. In addition, any forward-looking statements included in this press release represent the Company’s views only as of the date of its publication and should not be relied upon as representing its views as of any subsequent date. The Company specifically disclaims any intention to update any forward-looking statements included in this press release.
For Investors:
Bruce Mackle
LifeSci Advisors, LLC
+1-929-469-3859
bmackle@lifesciadvisors.com
For Media:
Brittany Leigh, Ph.D.
LifeSci Communications, LLC
+1-646-751-4366
bleigh@lifescicomms.com


FoxP3+ Regulatory T-Cells