- RT-111 achieved high bioavailability in humans -
- RT-111 was well-tolerated, with no serious adverse events -
- Celltrion has right of first negotiation to acquire worldwide rights to RT-111 following a Phase 1 clinical trial that meets its primary endpoints –
- Rani has now successfully completed three Phase 1 trials using RaniPill® technology -
- Company to host conference call today at
“We are highly encouraged by the positive results from our Phase 1 study for RT-111 – our third successfully completed Phase 1 trial using RaniPill® technology. To our knowledge, RT-111 is the first ever oral monoclonal antibody to achieve high bioavailability in humans,” said
Ustekinumab is a human IgG1қ monoclonal antibody that binds with specificity to the p40 protein subunit used by both the interleukin-12 and interleukin-23 (IL-12 and IL-23) cytokines. Currently, ustekinumab is available only as a subcutaneous injection (SC) and is marketed in
Rani’s single-center, open label, Phase 1 study of RT-111 was conducted in
Phase 1 Topline Results
Pharmacokinetics
- Oral RT-111 delivered ustekinumab biosimilar in a dose proportional manner and with high bioavailability (estimated at 84% relative to subcutaneous injection).
- Oral RT-111 demonstrated a higher Cmax and shorter Tmax compared to ustekinumab delivered by SC injection (0.5mg).
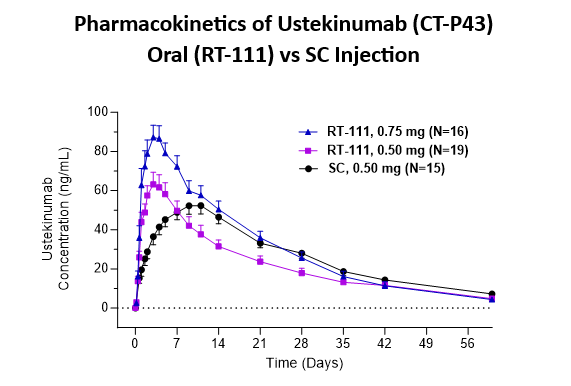
| Stelara® SC 0.50mg | RT-111 0.50mg | RT-111 0.75mg | |
| Cmax (ng/mL) | 56 ± 4 | 67 ± 7 | 92 ± 8 |
| Tmax (days) | 10 ± 0.8 | 3.1 ± 0.2* | 3.3 ± 0.2* |
| AUC (day*ng/mL) | 1,566 ± 130 | 1,315 ± 150 | 1,814 ± 165 |
Data are Mean ± SE from all subjects, including those with anti-drug antibodies. *p<0.0001 significantly different from SC group.
Safety and Tolerability
- RT-111 was well-tolerated by all participants in the two RT-111 groups, and no serious adverse events were observed in the study.
- There was no meaningful difference in incidence of anti-drug antibodies (ADA) via RaniPill® route of delivery, compared to STELARA® SC injection.
- No participants reported difficulty swallowing the capsule and capsule remnants passed from all participants without sequelae.
The ustekinumab biosimilar used in RT-111 is manufactured and supplied by Celltrion, Inc. (“Celltrion”) under Rani’s License and Supply Agreement with Celltrion announced in
Conference Call
Rani will host a corresponding conference call and live webcast at
About
Forward-Looking Statements
Statements contained in this press release regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements include statements regarding, among other things, the potential for RT-111 to be an alternative oral treatment option compared to burdensome injectables for autoimmune conditions, and the potential for Rani to make oral biologics a reality for millions of patients living with autoimmune conditions. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Words such as “intend” and “potential” and similar expressions are intended to identify forward-looking statements. These forward-looking statements are based upon Rani’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation, risks and uncertainties associated with Rani’s business in general and the other risks described in Rani’s filings with the
Investor Contact:
investors@ranitherapeutics.com
Media Contact:
media@ranitherapeutics.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/5b0b63b1-6b9a-4593-95cd-cbb9a0592101


Pharmacokinetics of Ustekinumab (CT-P43) Oral (RT-111) vs SC Injection
Pharmacokinetics of Ustekinumab (CT-P43) Oral (RT-111) vs SC Injection
2024 GlobeNewswire, Inc., source


