“MicroMatrix Flex has allowed me to expand how I use UBM technology in complex wound reconstruction procedures,” said Dr.
Integra’s UBM products – including MicroMatrix® UBM Particulate and Cytal® Wound Matrix sheets – have been used in more than 360,000 procedures over the past decade. During this period, UBM has been published in more than 200 pre-clinical and clinical publications. “We’re excited to be adding MicroMatrix Flex to our market-leading portfolio of dermal matrices,” commented
Dr.
For more information, visit www.MicroMatrixFlex.com.
About MicroMatrix Flex
MicroMatrix Flex is intended for the management of wounds including: partial and full-thickness wounds, pressure ulcers, venous ulcers, diabetic ulcers, chronic vascular ulcers, tunneled/undermined wounds, surgical wounds (donor sites/grafts, post-Mohs surgery, post-laser surgery, podiatric, wound dehiscence), trauma wounds (abrasions, lacerations, partial thickness burns, skin tears), draining wounds.
The device features a flexible dispensing tip to address tunneled, undermined, or irregular wounds. Its mixing system produces a paste with a flowable consistency designed to provide thorough contact with all areas of the wound.
About Integra
At
This news release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve risks and uncertainties that could cause actual results to differ from predicted results. Forward-looking factors that may be discussed include, but are not limited to, the commercial availability, market potential as well as potential therapeutic applications of the Integra products described herein. There can be no assurance these products will achieve the benefits described herein or that such benefits will be replicated. In addition, there can be no assurance that these products will be commercially successful. Forward-looking statements in this press release should be evaluated together with the many risks and uncertainties that affect Integra’s business and market, particularly those identified under the heading "Risk Factors" included in item 1A of Integra's Annual Report on Form 10-K for the year ended
Contacts:
Investors:
(609) 772-7736
chris.ward@integralife.com
Media:
(609) 208-8121
laurene.isip@integralife.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/c57f8724-df59-4e83-9899-45dae536fd07


MicroMatrix®Flex
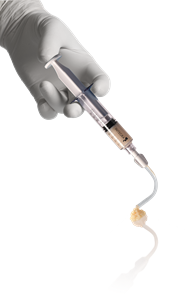
MicroMatrix®Flex is a dual-syringe system enabling the convenient mixing and precise delivery of MicroMatrix® paste.
2024 GlobeNewswire, Inc., source


