TNX-102 SL showed a robust effect size of 0.5 in improving fatigue and showed consistent activity across secondary measures of sleep quality, cognitive function, disability and Patient Global Impression of Change, but did not meet the primary endpoint of multi-site pain reduction at week 14
Findings fulfill the objectives of this proof-of-concept study, supporting the decision to advance the program based on a proposed primary endpoint using the PROMIS Fatigue scale
Tonix plans to meet with FDA to discuss a path to registration; fatigue is the signature symptom of Long COVID and the principal symptom overlapping with CFS/ME and fibromyalgia syndromes
PREVAIL was a randomized, double-blind, placebo-controlled, multi-site proof-of-concept study of 63 patients with laboratory-documented COVID-19 illness preceding Long COVID. PREVAIL was designed and conducted to guide the design of registrational studies of TNX-102 SL in fibromyalgia-type Long COVID. TNX-102 SL was generally well tolerated with an adverse event (AE) profile comparable to prior studies with TNX-102 SL. AE-related discontinuations were similar in drug and placebo arms. No new safety signals were observed.
“The robust activity of TNX-102 SL on the PROMIS Fatigue scale3-5 (ES=0.5, Figure 4) is important because patients and experts view fatigue as the signature symptom of Long COVID and it has been identified as the dominant symptom contributing to disability,”6 said Dr.
The Company intends to request an End-of-Phase 2 meeting with the
“The data from PREVAIL reinforce our belief in TNX-102 SL as a potential bedtime medicine for the management of fibromyalgia-type Long COVID,” said
Key Phase 2 PREVAIL Study Results
In the study, 63 subjects were enrolled and randomized 1:1 across approximately 30 U.S. sites to receive either TNX-102 SL or placebo daily at bedtime for 14 weeks. Subjects started with TNX-102 SL 2.8 mg tablet or one placebo tablet for the first 2 weeks and then increased to TNX-102 SL 5.6 mg (2 x 2.8 mg tablets) or two placebo tablets for the remaining 12 weeks of the treatment period. The percentage of subjects completing the study was 81.3% in the TNX-102 SL group and 80.6% in the placebo group. Demographics and baseline characteristics are shown in Table 1.
| Table 1: Demographics and Baseline Characteristics | |||
| Variable | Placebo | TNX-102 SL | Total |
| N=31 | N=32 | N=63 | |
| Age, mean years (SD) | 51.4 (10.01) | 48.6 (8.80) | 50.0 (9.45) |
| Female, number (%) | 25 (80.6%) | 21 (65.6%) | 46 (73.0%) |
| Male, number (%) | 6 (19.4%) | 11 (34.4%) | 17 (27.0%) |
| Ethnicity | |||
| Hispanic or Latino | 3 (9.7%) | 2 (6.3%) | 5 (7.9%) |
| Not Hispanic or Latino | 28 (80.6%) | 30 (93.8%) | 58 (92.1%) |
| Race | |||
| American Indian or AN, number (%) | 1 (3.2%) | 0 (0.0%) | 1 (1.6%) |
| Asian, number (%) | 0 (0.0%) | 1 (3.1%) | 1 (1.6%) |
| Black or | 5 (16.1%) | 7 (21.9%) | 12 (19.0%) |
| Native Hawaiian or PI, number (%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| White or Caucasian, number (%) | 24 (77.4%) | 21 (65.6%) | 45 (71.4%) |
| Multiple Races, number (%) | 1 (3.2%) | 3 (9.4%) | 4 (6.3%) |
| BMI, mean kg/m2 (SD) | 29.5 (4.44) | 29.8 (4.07) | 29.6 (4.22) |
| Employed, number (%) | 26 (83.9%) | 25 (78.1%) | 51 (81.0%) |
| Abbreviations: AN, | |||
Primary endpoint
- Given the lack of Long COVID treatments and the size of the current proof-of-concept study, an ES ≥ 0.2 was the pre-specified threshold for declaring the primary endpoint positive. The study trended towards a benefit but did not achieve statistical significance on the primary efficacy endpoint of change from baseline in the diary numerical rating scale (NRS) weekly average of daily self-reported worst Long COVID pain intensity scores for TNX-102 SL at the Week 14 endpoint versus placebo (effect size (ES) = 0.08, Figure 1.)
Secondary endpoints
- The change from baseline to the Week 14 endpoint for the daily sleep quality diary, PROMIS Sleep Disturbance, PROMIS Fatigue, PROMIS Cognitive function, the Insomnia Severity Index (ISI) and Sheehan Disability Scale showed numerical improvements (MMRM, ES ≥ 0.2): sleep diary (MMRM, ES =0.23, Figure 2.), PROMIS sleep Disturbance (MMRM, ES=0.32, Figure 3.), PROMIS fatigue (MMRM, ES=0.50, Figure 4.), PROMIS Cognitive Function -
Abilities , (MMRM, ES=0.21), the ISI (ANCOVA, ES=0.24) and the Sheehan Disability Scale (ANCOVA, ES=0.26). Moreover, robust activity was observed in the PGIC responder (very much improved or much improved) rate for TNX-102 SL compared to placebo (Figure 5): week 6 (31.3% vs. 9.7%, difference=21.6%), week 10 (28.1% vs. 12.9%, difference=15.2%), week 14 (34.4% vs. 16.1%, difference=18.2%).
Safety profile
TNX-102 SL demonstrated a favorable safety and tolerability profile over 14 weeks of treatment with no new safety signals. The most common adverse events are shown in Table 2. Participants with at least one treatment-emergent adverse event (TEAE) were at a rate of 56.3% on TNX-102 SL and 38.7% on placebo. In the TNX-102 SL group, 6.3% discontinued due to TEAE compared to 9.7% on placebo. Only one TEAE in the study was rated as severe, gastritis in a participant in the TNX-102 SL group. There were no serious adverse events (SAEs) in the study.
| Table 2: Adverse Events Occurring in ≥ 2 Participants in | ||||
| Placebo | TNX-102 SL | Total | ||
| N=31 | N=32 | N=63 | ||
| Administration Site Reactions | ||||
| Hypoaesthesia oral | 0 | 6 | 6 | |
| Product taste abnormal | 0 | 3 | 3 | |
| Glossodynia | 0 | 2 | 2 | |
| Oral pain | 0 | 2 | 2 | |
| Paraesthesia oral | 0 | 2 | 2 | |
| Systemic Adverse Events | ||||
| Influenza like illness | 2 | 0 | 2 | |
Abbreviations: LS, least squares; SE, standard error
Abbreviations: LS, least squares; SE, standard error
Abbreviations: LS, least squares; SE, standard error; SD, sleep disturbance
Abbreviations: LS, least squares; SE, standard error
*p=0.034, #p=0.096
About the Phase 2 PREVAIL Study
The Phase 2 PREVAIL proof-of-concept study was a 14-week double-blind, randomized, multicenter, placebo-controlled study to evaluate the efficacy and safety of TNX-102 SL taken daily at bedtime in patients with multi-site pain associated with laboratory-documented post-acute sequelae of SARS-CoV-2 infection (PASC). The trial was conducted at approximately 30 sites in the
For more information, see ClinicalTrials.gov Identifier: NCT05472090.
About Long COVID or Post-Acute Sequelae of COVID-19 (PASC)
Post-acute sequelae of COVID-19, or PASC is the formal name for a condition now widely known as Long COVID. The
About Fibromyalgia-Type Long COVID
Common symptoms of Long COVID, including multi-site pain, fatigue, unrefreshing sleep, and cognitive dysfunction, or ‘brain fog,’ are hallmarks of conditions like fibromyalgia and CFS/ME. Defining subgroups of Long COVID patients that overlap with fibromyalgia and CFS/ME is expected to facilitate the development of new treatments.20 We are studying TNX-102 SL in the subgroup of Long COVID patients whose symptoms overlap with fibromyalgia, which we have termed ‘Fibromyalgia-type Long COVID.’ TNX-102 SL is in phase 3 development for the management of fibromyalgia.8,21 Fibromyalgia has been recognized by the
About TNX-102 SL
TNX-102 SL is a patented sublingual tablet formulation of cyclobenzaprine hydrochloride which provides rapid transmucosal absorption and reduced production of a long half-life active metabolite, norcyclobenzaprine, due to bypass of first-pass hepatic metabolism. As a multifunctional agent with potent binding and antagonist activities at the 5-HT2A-serotonergic, α1-adrenergic, H1-histaminergic, and M1-muscarinic receptors, TNX-102 SL is in development as a daily bedtime treatment for fibromyalgia, Long COVID (formally known as post-acute sequelae of COVID-19 [PASC]), alcohol use disorder and agitation in Alzheimer’s disease. The United States Patent and Trademark Office (USPTO) issued United States Patent No. 9636408 in
*TNX-102 SL is an investigational new drug and is not approved for any indication
Citation:
- NIH News Release.
July 31, 2023 . “NIH launches long COVID clinical trials through the RECOVER Initiative, opening enrollment.” https://www.nih.gov/news-events/news-releases/nih-launches-long-covid-clinical-trials-through-recover-initiative-opening-enrollment (accessedSeptember 4 , 2023). - Johnson, M and Goldstein A.
Washington Post .July 31, 2023 . “NIH announces long covid treatment studies with hundreds of patients” www.washingtonpost.com/health/2023/07/31/long-covid-treatment-studies-nih/ - Cook, K.F., et al. 2016
Journal of Clinical Epidemiology , 73, 89- 102 - Cella, D., et al. 2016
Journal of Clinical Epidemiology , 73, 128–134 - Lai, J.S., et al. 2011Archives of Physical Medicine and Rehabilitation, 92(10 Supplement), S20-S27.
- Walker S, et al. BMJ Open 2023;13:e069217. doi:10.1136/ bmjopen-2022-069217
- Thaweethai T, et al. JAMA. 2023. 329(22):1934-1946
- Lederman S, et al. Arthritis Care Res. 2023 https://doi.org/10.1002/acr.25142
- Sullivan, G et al. Tonix data on file, 2023
- NIH Press Release.
July 31, 2023 . “HHS Announces the Formation of theOffice of Long COVID Research and Practice and Launch of Long COVID Clinical Trials Through the RECOVER Initiative” www.hhs.gov/about/news/2023/07/31/hhs-announces-formation-office-long-covid-research-practice-launch-long-covid-clinical-trials-through-recover-initiative.html (accessedSeptember 4 , 2023). - NIH News Release.
June 22, 2022 . “Nearly One in Five American Adults Who Have Had COVID-19 Still Have ‘Long COVID’” https://www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/20220622.htm (accessedSeptember 4 , 2023). - Briggs, A, and Vassall, A. Nature. 2021. 593(7860): 502-505
- Nittas V, et al. Public Health Rev. 2022. 43:1604501
- Davis, HE., et al. EClinicalMedicine. 2021. 38:101019
- Martin C, et al. PLoS One. 2021. 16(12):e0260843
- The
NIH provision ofTitle III Health and Human Services, Division M--Coronavirus Response and Relief Supplemental Appropriations Act, 2021, of H.R. 133, The Consolidated Appropriations Act of 2021. The bill was enacted into law on27 December 2020 , becoming Public Law 116-260. Department of Health and Human Services ,Office of the Assistant Secretary for Health . 2022. National Research Action Plan on Long COVID,200 Independence Ave SW ,Washington, DC 20201. www.covid.gov/assets/files/National-Research-Action-Plan-on-Long-COVID-08012022.pdf- Maixner W, et al. J Pain. 2016. 17(9 Suppl):T93-T107
- Haider S, et al. Pain. 2023. 164(2):385-401
- Sutherland, S.
Scientific American . 2023 - Tonix Pharmaceuticals Press Release.
Feb 22, 2023 . https://ir.tonixpharma.com/news-events/press-releases/detail/1369/tonix-pharmaceuticals-describes-emerging-research-on-the - Moldofsky H, et al. J Rheumatol. 2011. 38(12):2653-63
- Clauw DJ, and Calabrese L. Ann Rheum Dis. 2023
- Clauw DJ, et al. Pain. 2020. 161(8):1694-1697
Tonix is a biopharmaceutical company focused on commercializing, developing, discovering and licensing therapeutics to treat and prevent human disease and alleviate suffering. Tonix Medicines, our commercial subsidiary, markets Zembrace® SymTouch® (sumatriptan injection) 3 mg and Tosymra® (sumatriptan nasal spray) 10 mg under a transition services agreement with
*Tonix’s product development candidates are investigational new drugs or biologics and have not been approved for any indication.
Zembrace SymTouch and Tosymra are registered trademarks of Tonix Medicines. Intravail is a registered trademark of
This press release and further information about Tonix can be found at www.tonixpharma.com.
Forward Looking Statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. These statements may be identified by the use of forward-looking words such as “anticipate,” “believe,” “forecast,” “estimate,” “expect,” and “intend,” among others. These forward-looking statements are based on Tonix's current expectations and actual results could differ materially. There are a number of factors that could cause actual events to differ materially from those indicated by such forward-looking statements. These factors include, but are not limited to, risks related to the failure to obtain FDA clearances or approvals and noncompliance with FDA regulations; risks related to the failure to successfully market any of our products; risks related to the timing and progress of clinical development of our product candidates; our need for additional financing; uncertainties of patent protection and litigation; uncertainties of government or third party payor reimbursement; limited research and development efforts and dependence upon third parties; and substantial competition. As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products. Tonix does not undertake an obligation to update or revise any forward-looking statement. Investors should read the risk factors set forth in the Annual Report on Form 10-K for the year ended
Investor Contact
investor.relations@tonixpharma.com
(862) 904-8182
ICR Westwicke
peter.vozzo@westwicke.com
(443) 213-0505
Media Contact
ICR Westwicke
ben.shannon@westwicke.com
(919) 360-3039
Photos accompanying this announcement are available at https://www.globenewswire.com/NewsRoom/AttachmentNg/07482efe-4b16-4cb5-a5d8-1608e1e5412c
https://www.globenewswire.com/NewsRoom/AttachmentNg/e16dda0e-ab4a-44aa-aeb5-919ce14afb84
https://www.globenewswire.com/NewsRoom/AttachmentNg/bbe66a9f-1154-4117-a045-4d07e061660d
https://www.globenewswire.com/NewsRoom/AttachmentNg/18539d75-14de-4e41-800a-5d2ff547f9d3
https://www.globenewswire.com/NewsRoom/AttachmentNg/de06c2b0-02b7-4efa-9f67-ea4ef1429b63


Figure 1.
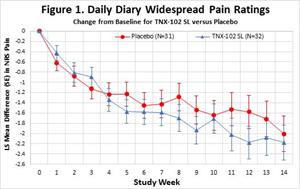
Daily Diary Widespread Pain Ratings
Figure 2.
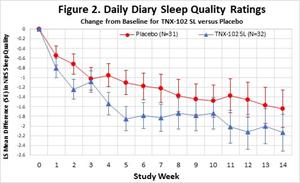
Daily Diary Sleep Quality Ratings
Figure 3.
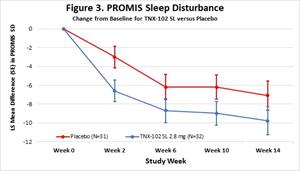
PROMIS Sleep Disturbance
Figure 4.
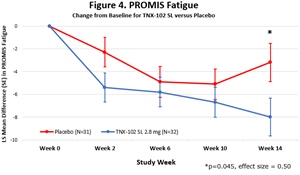
PROMIS Fatigue
Figure 5.
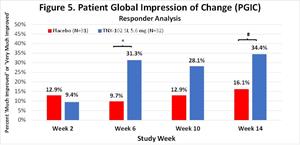
Patient Global Impression of Change (PGIC)
2023 GlobeNewswire, Inc., source

