The inception of this study highlights Acutus’ unique therapeutic position offering the first and only force sensing gold-tip ablation catheter, AcQBlate Force, designed to deliver either radiofrequency (RF) or PFA therapy – with the added benefit of fully integrating with therapy guidance provided by the novel AcQMap 3D Imaging and Mapping System.
“The vision for pioneering Pulsed Field Ablation treatment was primarily due to PFA’s ability to have high-impact treatment while preserving healthy tissue. As physicians, we know that the majority of our patients that come back for re-treatment following ablation for atrial fibrillation have triggers in various locations beyond the standard course of care, and it’s imperative for us to research beyond pulmonary vein isolation-only utilizations,” said Dr.
“Pulsed Field Ablation as applied to the field of cardiac electrophysiology has enormous potential, given the expected improvements in safety, efficacy and speed over current therapies,” said
About Acutus’ RF Ablation System for Electrophysiology Procedures
Acutus’ therapeutic radiofrequency ablation system and force sensing catheter is currently approved and commercially available for use in
About Pulsed Field Ablation
Contemporary Pulsed Field Ablation technologies utilize short bursts of high voltage microsecond or nanosecond electrical pulses to selectively isolate areas of cardiac tissue that are thought to contribute to triggering or sustaining cardiac arrhythmias. PFA is currently being investigated as an alternative to cryo and radiofrequency ablations, which ablate tissue by either extreme cold or extreme heat. When designed and applied appropriately, the therapeutic treatment effect of pulsed field ablation energy is almost exclusively confined to the targeted cardiac tissue without raising or lowering tissue temperature. Focal PFA may allow for more precise demarcated lesions, promoting rapid tissue healing and facilitating practical safety margins for nearby tissue structures outside the heart.
AcQForce PFA-CE Study
AcQForce PFA-CE is a prospective, non-randomized European clinical study designed to demonstrate the safety and performance of the Acutus Pulsed Field Ablation system in up to 60 patients undergoing ablation therapy for AF.
Site recruitment and patient enrollment for AcQForce PFA-CE is ongoing. For more information, please visit https://www.clinicaltrials.gov reference NCT05113056.
*The AcQBlate Force Sensing System is CE marked for RF energy delivery. The AcQBlate Force Sensing System is an investigational device and is limited by
About
To learn more about Acutus Medical’s complete portfolio of diagnostic, access and therapy products, please visit https://acutusmedical.com.
Follow
US Media Contacts
(619) 929-1275
holly.windler@acutus.com
Levitate
(260) 408-5383
acutus@levitatenow.com
Investor Contact
(415) 202-5678
caroline.corner@westwicke.com
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/272a48bb-8eee-40c4-9d3c-ffd6f23f768a
https://www.globenewswire.com/NewsRoom/AttachmentNg/b2ce8bad-6540-4108-8c97-119201b32ad3

![]()
Acutus Medical’s Electrophysiology System
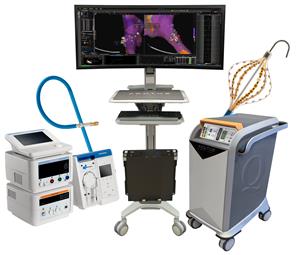
AcQForce PFA-CE study is designed to demonstrate the safety and performance of the Acutus focal PFA system utilizing the AcQBlate Force Sensing Ablation Catheter (pictured) and system (not shown) and AcQMap 3D mapping system (pictured) in patients undergoing ablation therapy for atrial fibrillation.
Acute Pulmonary Vein Isolation Confirmed Utilizing Acutus Technology with SuperMap
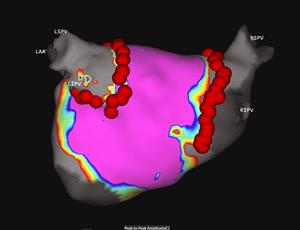
Acutus SuperMap image shows acute pulmonary vein isolation in the first patient enrolled in the AcQForce PFA-CE clinical study. Circumferential ablation lesions were delivered with Acutus Pulsed Field Ablation using Acutus’ gold-tip contact force sensing ablation catheter, AcQBlate Force. Imaging and confirmation of the acute pulmonary vein isolation were performed with the AcQMap non-contact mapping system.
2021 GlobeNewswire, Inc., source

