Phase 3 Data Collected to Date Sufficient for Submission of New Drug Application
CTx-1301 Could Benefit >60% of ADHD Patients Currently Using Immediate-Release Booster Doses
Advancing Discussions with
Ready to Engage with Payers for Reimbursement and Market Access
Details from the FDA communication to
- Per the 505(b)(2) pathway, submission of CTx-1301’s NDA does not require continuation of the fixed dose study and the onset and duration study;
- Data collected to date from these two Phase 3 studies will be included in the filing; and
- Contemporaneously with the preparation of the NDA filing,
Cingulate will conduct a Phase 1 fed/fast study, the study is expected to last approximately two to three weeks, with the results being included in the NDA filing.
“We are pleased with the FDA’s guidance and perspective regarding our pathway to a timely NDA filing, and we will proceed to complete all remaining requirements accordingly,” said
“If approved,” Childress continued, “I believe CTx-1301 could potentially benefit the more than 60 percent of patients currently using immediate-release booster doses to extend the efficacy of their medications.”
Earlier this year,
The results were previously presented at the 2023
The
About Attention Deficit/Hyperactivity Disorder (ADHD)
ADHD is a chronic neurobiological and developmental disorder that affects millions of children and often continues into adulthood. The condition is marked by an ongoing pattern of inattention and/or hyperactivity-impulsivity that interferes with functioning or development. In the
About CTx-1301
Cingulate’s lead candidate, CTx-1301, utilizes Cingulate’s proprietary PTR drug delivery platform to create a breakthrough, multi-core formulation of the active pharmaceutical ingredient dexmethylphenidate, a compound approved by the FDA for the treatment of ADHD. Dexmethylphenidate is part of the stimulant class of medicines and increases norepinephrine and dopamine activity in the brain to affect attention and behavior. While stimulants are the gold standard of ADHD treatment due to their efficacy and safety, the long-standing challenge continues to be providing patients with an entire active-day duration of action. CTx-1301 is designed to precisely deliver three releases of medication at the predefined time, ratio, and style of release to optimize patient care in one tablet. The result is a rapid onset and entire active-day efficacy, with the third dose being released around the time when other extended-release stimulant products begin to wear off.
About Precision Timed Release™ (PTR™) Platform Technology
About
Forward-Looking Statements
This press release contains “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements include all statements, other than statements of historical fact, regarding our current views and assumptions with respect to future events regarding our business, including statements with respect to our plans, assumptions, expectations, beliefs and objectives with respect to product development, clinical studies, clinical and regulatory timelines, market opportunity, competitive position, business strategies, potential growth opportunities and other statements that are predictive in nature. These statements are generally identified by the use of such words as “may,” “could,” “should,” “would,” “believe,” “anticipate,” “forecast,” “estimate,” “expect,” “intend,” “plan,” “continue,” “outlook,” “will,” “potential” and similar statements of a future or forward-looking nature. Readers are cautioned that any forward-looking information provided by us or on our behalf is not a guarantee of future performance. Actual results may differ materially from those contained in these forward-looking statements as a result of various factors disclosed in our filings with the
Investor Relations
Vice President, Investor & Public Relations,
TDalton@cingulate.com
913-942-2301
mkreps@darrowir.com
214-597-8200
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/00c8b334-57ec-49cf-9a59-02d75463089e


Cingulate's Precision Timed Release Technology™ (PTR™) Platform Technology
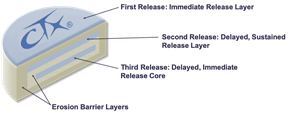
Cingulate® is developing ADHD product candidates capable of achieving true once-daily dosing using the Company’s innovative Precision Timed Release™ (PTR™) drug delivery platform technology incorporating a proprietary Erosion Barrier Layer (EBL) that provides control of drug release at precise, pre-defined times with no release of drug prior to the intended release.The technology is an erodible barrier layer that is wrapped around a drug containing core to give a tablet-in-tablet dose form. The barrier layer is designed to erode at a controlled rate until eventually the drug is released from the core tablet.CTx-1301 (dexmethylphenidate) and CTx-1302 (dextroamphetamine) have pioneering compression technology with specialized release characteristics that may enable patients to take one tablet per day and avoid the use of additional immediate release “boosters.”We believe our most advanced drug product candidate, CTx-1301, will be the first true once-daily dexmethylphenidate tablet
2024 GlobeNewswire, Inc., source


