- The expansion trial is expected to enrol 10 patients with bile tract cancers (cholangiocarcinoma)
- Interim results from the MAST trial have demonstrated positive responses in gastrointestinal cancers, particularly in cholangiocarcinoma where one patient treated with CF33-hNIS (VAXINIA) achieved a complete response and another patient achieved stable disease
- The fifth cohort in the intratumoural (IT) arm of the VAXINIA monotherapy trial has now been cleared with no safety signals seen to date; the cohort included patients with thymic carcinoma, triple negative breast cancer and cholangiocarcinoma
Imugene Managing Director & CEO
The expansion of the MAST (Metastatic Advanced Solid Tumours) Phase 1 trial is planned for 10 patients with bile tract cancers, after early positive responses were observed in gastrointestinal cancers, particularly in cholangiocarcinoma. Cholangiocarcinoma is a rare disease in which malignant cancer cells form in the bile ducts. It is difficult to treat and generally responds poorly to immunotherapy drugs.
One patient with cholangiocarcinoma who had failed three prior lines of therapy received a mid-dose of IT-administered monotherapy VAXINIA achieved a complete response, meaning the disappearance of all signs of cancer in response to treatment, with no known recurrence in more than 430 days. A second patient with cholangiocarcinoma, who has also progressed on prior drug therapies, achieved stable disease for more than four months upon receiving IV-administered VAXINIA.
In
On Friday
The multicenter, Phase 1, MAST trial commenced by delivering a low dose of VAXINIA to patients with metastatic or advanced solid tumours who have had at least two prior lines of standard of care treatment. With no safety signals identified to date, the trial has since progressed through the monotherapy dose escalation cohorts as well as the combination study, whereby VAXINIA is administered with well-known checkpoint inhibitor pembrolizumab. CF33 oncolytic virus, developed by City of Hope, has been shown to shrink colon, lung, breast, ovarian and pancreatic cancer tumours in preclinical laboratory and animal models¹.
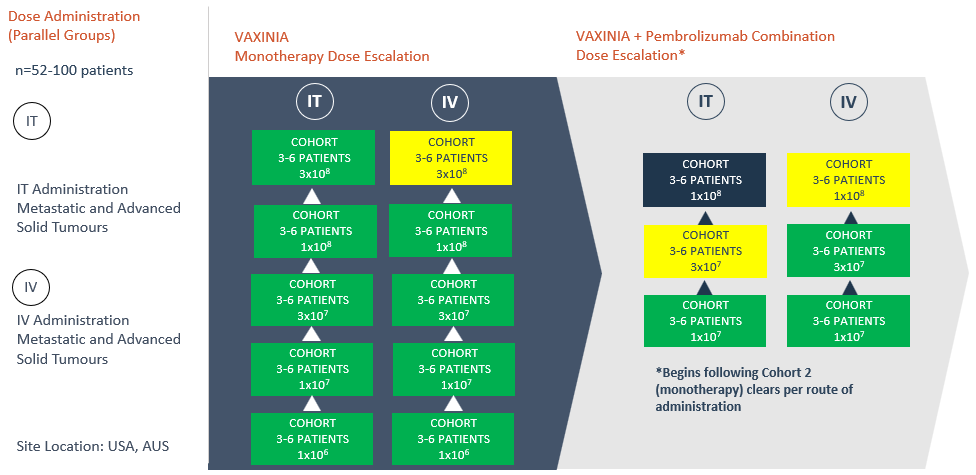
Further dose escalation to continue as long as no safety issues are observed
For more information please contact:
Managing Director and Chief Executive Officer
info@imugene.com
Investor Enquiries
shareholderenquiries@imugene.com
US Investor and Media Enquiries
harmstrong@imugene.com
Media Enquiries
matt@nwrcommunications.com.au
Connect with us on LinkedIn @Imugene Limited
Follow us on Twitter @TeamImugene
Watch us on YouTube @ImugeneLimited
References
¹ Warner SG, Kim SI, Chaurasiya S, O'Leary MP, Lu J, Sivanandam V, Woo Y, Chen NG, Fong Y. A Novel Chimeric Poxvirus Encoding hNIS Is Tumor-Tropic, Imageable, and Synergistic with Radioiodine to Sustain Colon Cancer Regression. Mol Ther Oncolytics. 2019 Apr 11;13:82-92. doi: 10.1016/j.omto.2019.04.001. PMID: 31061881; PMCID: PMC6495072.
About
Our vision is to help transform and improve the treatment of cancer and the lives of the millions of patients who need effective treatments. This vision is backed by a growing body of clinical evidence and peer-reviewed research.
Release authorised by the Managing Director and Chief Executive Officer Imugene Limited.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/24a0253d-936d-4df5-8df2-02ea3355f179

Imugene’s Phase 1 CF33-hNIS (VAXINIA) MAST Trial
Enrolment has opened for Imugene’s expansion study in bile tract cancer (cholangiocarcinoma) patients, having completed the fifth, high dose cohort in the intratumoural (IT) arm of the monotherapy dose escalation study.
2024 GlobeNewswire, Inc., source



