The first results of Novo Nordisk's Select study, unveiled in August, revealed that Wegovy, which helps patients lose an average lose an average of 15% of their body weight, also reduces the occurrence of heart attacks, strokes and heart disease-related deaths by 20%. The full findings of the study, presented at the American Heart Association's Annual Scientific Meeting in Philadelphia and published in the New England Journal of Medicine, suggest that the drug has other health benefits beyond those known from weight loss. Investors were not mistaken: the stock gained 3.4% to DKK 715 during trading on Monday November 13.
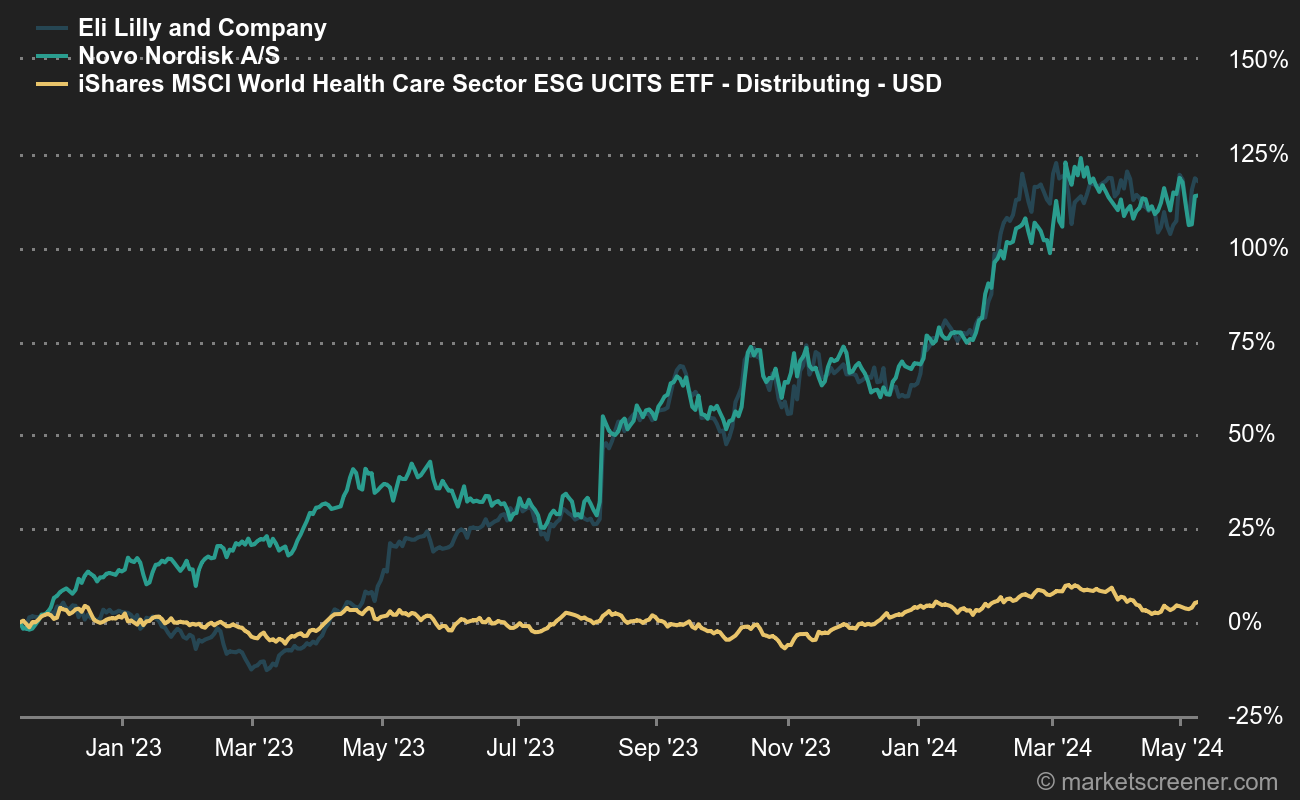
A positive additional effect
According to the researchers, the difference in cardiac risk between patients given Wegovy, known chemically as semaglutide, and those given a placebo appeared almost immediately after the start of treatment. In the study of patients who were overweight or obese according to body mass index and had pre-existing heart disease but no diabetes, Wegovy reduced the risk of non-diabetic heart attack.Wegovy reduced the risk of non-fatal heart attack by 28%, non-fatal stroke by 7% and cardiac death by 15% compared with placebo.
The fact that patients had not started losing weight when the cardiovascular benefits first became apparent suggests that heart protection was not solely the result of weight loss, said Novo. The researchers said that while understanding the mechanisms of cardiovascular protection by semaglutide remains hypothetical, there was a cohesive effectrent effect on associated risk factors that supports the idea that multiple pathways are behind the drug's clinical benefit.
Patients on Wegovy experienced a reduction in C-reactive protein, an indication of inflammation, similar to that seen with cholesterol-lowering statins, which are known to significantly reduce cardiac risk, the researchers reported. "The cardiovascular benefit (of Wegovy) is a combination of several factors, but I would cite glycemic control, weight loss and inflammation," explained Martin Lange, head of development at Novo Nordisk, in an interview.
According to the study, in the trial involving 17,604 patients and lasting an average of 33 months, nearly 1,500 patients taking Wegovy discontinued treatment due to adverse side effects, mainly gastrointestinal disorders such as nausea and vomiting, compared with 718 patients in the placebo group. Although the trial was not conducted to test weight loss, participants lost an average of almost 10% of their total body weight. Novo said that patients in the cardiac study were not required to follow their diet and exercise regimen, as is the case in obesity trials. The study showed that Wegovy was safe and well tolerated, in line with previous semaglutide trials, Novo said.
Towards updating the Wegovy label to include heart benefits
Lange said he expects the application to update Wegovy's label to include heart benefits to be approved in the US in the first half of next year and in the European Union in the second half. Drug regulatory authorities can update drug label information to include new data or reflect new indications for use after initial approval.
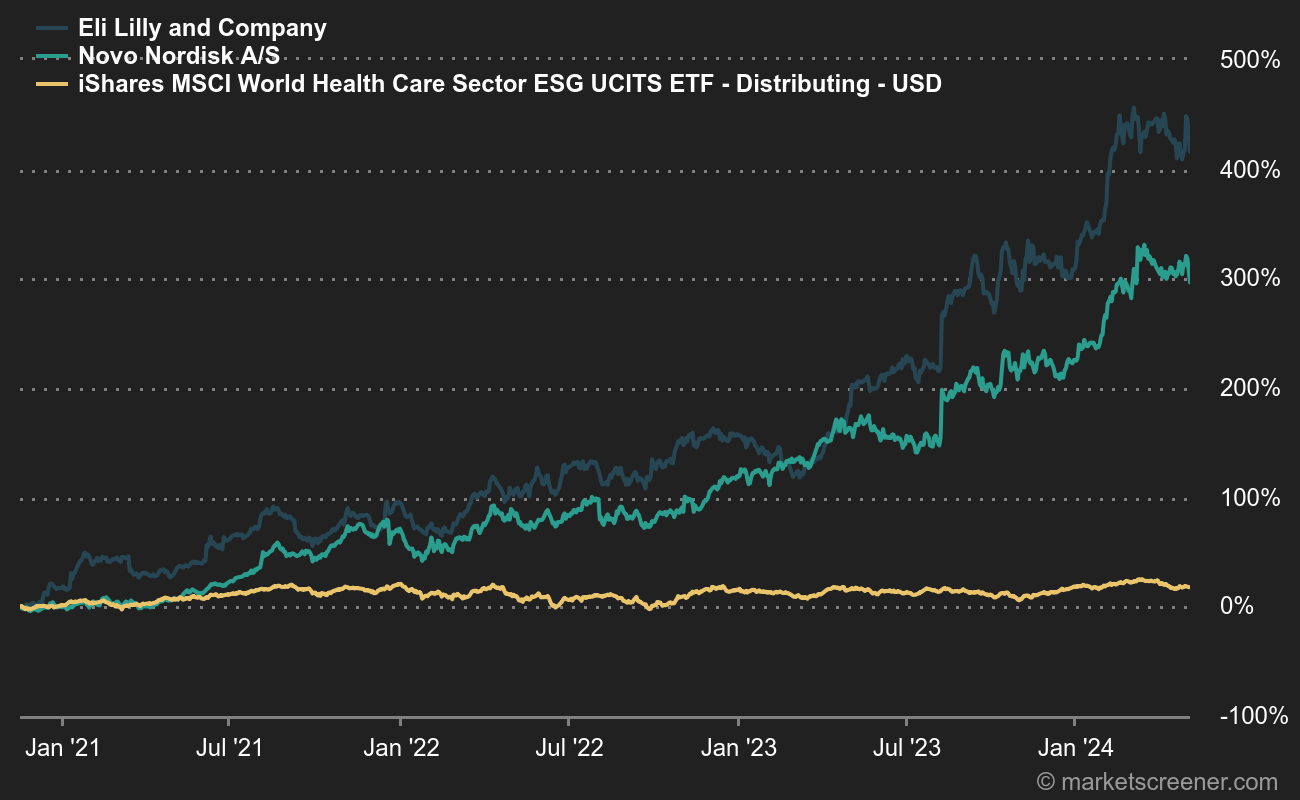
Lilly and Novo's three-year outperformance is impressive
Novo Nordisk reported last week that the US Food and Drug Administration had accepted its request for a label update under a priority review, meaning that the agency will make a decision within six months. On Wednesday, the American and British authorities approvedEli Lilly's rival weight-loss treatment, which had already been approved and marketed under the name Mounjaro for diabetes. These two laboratories have been the real stars of the pharmaceutical sector in recent years.

 By
By 























